|
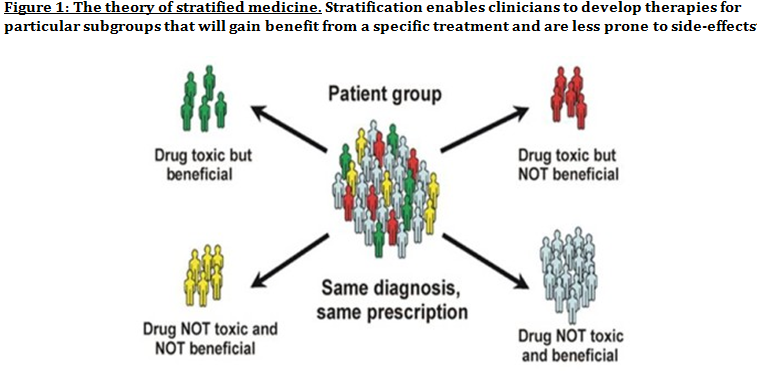
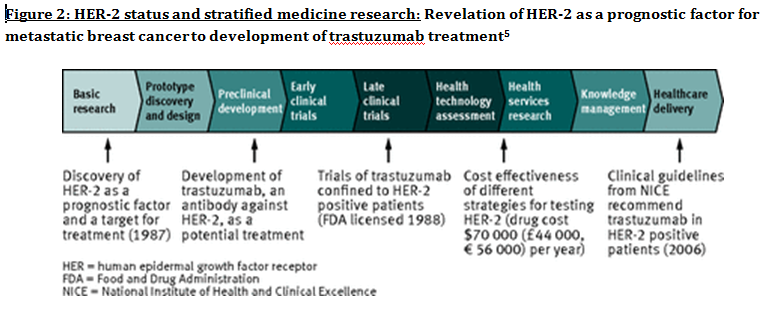
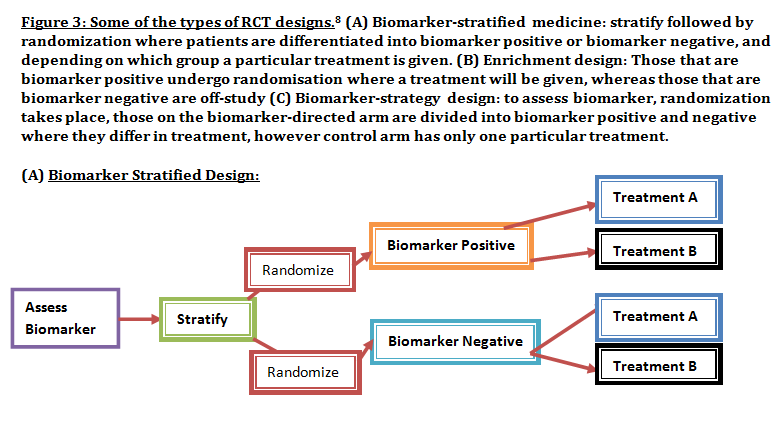
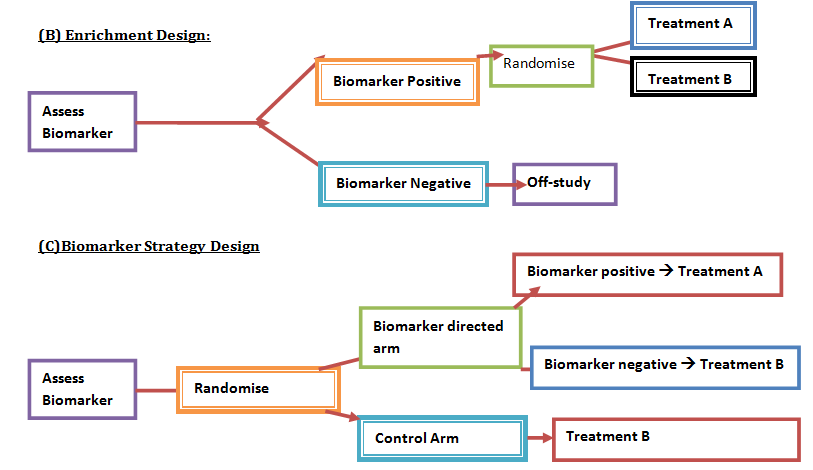
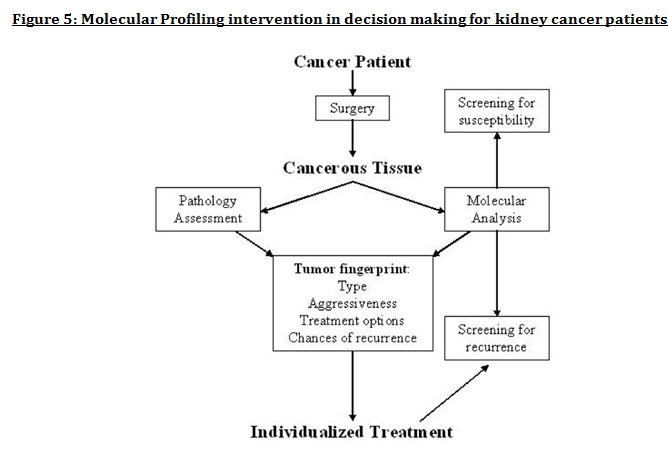
Advanced understanding in the molecular aspect of disease generated progress leading to a novel healthcare approach called stratified medicine. From recent years it transitioned from several stratified medicines such as Imanitib, a mutant kinase inhibitor prescribed for patients with chronic myeloid leukaemia to a variety of medicines today such as Zelboraf; targets BRAF proteins in patients with V600E mutation in melanoma. Stratified medicine is defined as a treatment derived from utilizing molecular or genetic information to discover patients with different mechanisms of disease or a specific therapeutic response as illustrated in Figure 1. This allows identification and development of therapies and diagnostic tools that give better health stances such as safety and effectiveness, for an aimed sub-group of patients that have analogous biological characteristics. Besides patients, there are also benefits for clinicians, economics and healthcare system. Thus, stratification plays a significant role in medicine especially cancer therapy. One of the ways in which stratified medicine alters cancer treatment can be seen through HER-2 status in breast cancer management illustrated in Figure 2. This is where findings from prognosis research are translated towards developing better patient outcomes. At first, HER-2 protein was found as a prognostic factor due to overexpression in breast cancer cells. This led to development of Trastuzumab (Herceptin) as a potential treatment against HER-2. Then, evaluation in trials took place where subjects recruited were women with HER-2 positive cancers. Due to successful results, Herceptin was prescribed to HER-2 positive patients and not those that were HER-2 negative. Those that were negative had alternative treatments. Another way in which stratified medicine is altering cancer treatment is via biomarkers. Biomarkers are defined as biological characteristics that give an indication of pathogenic process, normal biological state or pharmacological response to a therapeutic involvement. They can be cellular, biochemical or genetics. These markers allow identification of sub-groups that are able to respond positively or negatively due to a particular treatment. A number of randomised control designs (RCT) are used to assess predictive biomarkers such as biomarker-strategy design presented in Figure 3A. They are utilized in scenarios where there are two or more therapeutic options8. Patients are assigned on a random basis despite of their biomarker status8. An analysis plan is then stratified via biomarker to examine the treatment effect. An example of this is Marker Validation for Erlotinib in Lung Cancer (MARVEL) trial8. Patients with non-small cell lung cancer (NSCLC) are allocated to either treatment: Pemetrexed or Erlotinib. The analysis plan is then stratified via epidermal growth factor receptor gene (EGFR) status. This is followed by qualitative analysis via fluorescent in situ hybridization (FISH). Advantages of using biomarker-strategy design are that it examines relative efficacy of each sub-group. However, it is unreasonable for multifaceted therapeutic approaches and several treatments maybe inappropriate for some biomarker groups. Another type of RCT design is Enrichment design (Figure 3B), it consists of evaluating all patients, however the analysis plan is stratified to specific patients with definite biomarker values8. For example in CALGB-10603 trial, utilized a predictive biomarker allowing only acute myeloid leukaemia patients with FLT3 mutation. They were allocated randomly to a normal treatment or a treatment that entailed FLT3 kinase inhibitor; midostaurin, whereas, those without FLT3 mutation where ‘off-study’. One of the benefits of using enrichment design is that it utilizes a small sample. Nevertheless, it requires potent biological basis and the biomarker may not identify correctly the sub-group that would gain. Additionally, biomarker-strategy design (Figure 3C) entails patients who are allocated randomly and uses an investigational treatment arm that utilizes biomarker to direct treatment or to a control arm that does not direct treatment8. An example of this can be seen with Excision repair cross-complementing 1 (ERCC1) as biomarker. It is linked to cisplatin resistance in NSCLC. Therefore, in the trial patients are allocated on a random basis to the control arm8. This is where they will be given two treatments: docetaxel and cisplatin8. However patients may be allocated to the biomarker-strategy arm where patients are altered to gemcitabine and docetaxel treatment rather than being categorized as cisplatin resistant. Patients thare are not resistant to cisplatin undergo the standard treatment which is composed of cisplatin and docetaxel. This design can be used in complex therapeutic approaches. On the contrary, a positive trial does not demonstrate biomarker use. This emphasises the clinical relevance of biomarkers towards cancer treatment. However, there are disadvantages of utilizing biomarkers such as insufficient handling, processing and storage of samples. This can lead to their degradation and false results. For improvement, removal of quality samples for biomarker analysis from reliable sources reduces issues to patients. This will need design and validation of protocols for procedures. A technique in which cancers can be stratified is genomic analysis using DNA and RNA microarrays. Microarrays consist of RNA or DNA pieces from genes and quantify the amount of corresponding DNA or RNA in the provided sample. Therefore, genetic variants or expression levels of disease-related genes can be observed. For example MammaPrint, a microarray-based diagnostic technique can examine genetic variants in breast cancer tissue to estimate possibility of metastasis. However, microarrays are limited because they can only assess genes presented there. DNA microarrays will potentially be substituted with whole genome sequencing due to benefit of obtaining complete information, whereas RNA microarrays remains valuable for exploring gene expression. Moreover, metabolomic and protein analyses are another method used to stratify patients. Microarrays can be utilized where antibodies fuse to desired metabolite or protein variant. In comparison to genomics, both analyses provide more information on molecular mechanisms involved in a disease. A combination of their data, genomics and clinical outcomes indicates the impact of biomedical and health informatics on progress in stratified medicine. Nevertheless, storage conditions can affect results despite extraction of molecules can be easily obtained. Other techniques by which cancers maybe stratified are immunohistochemistry and polymerase chain reaction (PCR). They are routinely used in contrast to genomic, proteomic or metabolomic analysis. Immunohistochemistry utilizes antibodies that bind to a specific molecule. This is normally a cell-surface protein and appears with a unique colour under microscopic analysis. This technique can be performed using oestrogen receptor biomarker in breast cancer. Another example is PD-1 ligand in directing antibody therapy for patients with colorectal cancer and melanoma. Nonetheless, immunohistochemistry performed in clinical setting, diagnostic-grade antibodies are not used always. In relation to qPCR, they examine a desired DNA sequence in the sample that are normally cancer mutations. Similar to immunohistochemistry, they do not always perform using standardised processes. Thus, prospective techniques such as X-ray computed tomography, positron-emission tomography (PET) and magnetic resonance imaging do not require invasive procedures to obtain clinical samples. To observe the patient’s interior body, one examines what happens between electromagnetic radiations with the body. 18-fluorodeoxyglucose (FDG), a PET-active glucose analogue that resides in malignant tumour tissues with enhanced metabolic rate. In combination with PET, FDG is useful for diagnosis and examining therapeutic response in many types of cancer such as lymphomas. From a bedside perspective, molecular profiling appears to be a potential technique in identifying biomarkers in renal cell carcinoma aiding in decision-making for kidney cancer patients illustrated in Figure 5. Molecular profile is an analysis of protein and gene expression and leads to developing novel targeted therapeutic response. Furthermore, mutational stratification aids clinicians to determine which individuals will respond to which particular treatment. For instance, identifying KRAS in bowel cancer, Cetuximab and Panitumumab are observed to see if it implicated. Another example of how mutational stratification is likely to alter cancer treatment is gastrointestinal stromal tumour(GIST). Patients with GIST are prescribed with Imatinib. Some patients benefited where there was low GIST recurrence rates whereas others did not. Recent studies have demonstrated how mutational analysis is a predictive substance of recurrence-free survival. Patients with mutations in exon 11 of KIT gene, had more recurrence-free survival in comparison to patients with exon 9. Patients with wild-type GIST generally had disease but narrow sensitivity to Imatinib in most cases. Thus, risk-stratification and mutational analysis is necessary for optimising therapeutic approaches.
Lung cancer is another example of where mutational stratification has been used. It is the most common cause of mortality globally. According to statistics, 70% of NSCLC patients have durable effect and limited treatment obtained where 50% of them have an unknown cause. By using a specific genetic dependency screen one would be able to recognise vital somatic mutations. Findings discovered 3 kinases that have gain-in-function mutations in lung cancer mediating towards ERK pathway: PAK5, FGFR4 and MAP3K9. Consequently, mutated kinases prevent proliferation, inhibit downstream signalling events and eradicate lung cancer cells. Ultimately, stratified medicine plays a significant role in cancer treatment. Researchers are currently identifying more biomarkers to improve potential treatments. Cancer Research UK’s Stratified Medicine Programme entails molecular basis information of 9,000 tumours. It is co-working with pharmaceutical companies such as Pfizer to develop a national service model to routinely examine tumours for particular genetic variations. This will help clinicians with decision–making and aid researchers on how particular variations influence specific treatments.
3 Comments
3/21/2015 05:29:15 pm
Thus, stratification plays a significant role in medicine especially cancer therapy.
Reply
8/9/2016 01:06:21 am
Another way in which stratified medicine is altering cancer treatment is via biomarkers
Reply
5/11/2022 01:31:21 am
I very much appreciate it. Thank you for this excellent article. Keep posting!
Reply
Leave a Reply. |
Details
This project began as a facebook page sharing information about different illnesses, diagnosis and treatments. We are now doing short articles :)
Health stuffArchives
April 2020
Categories |
 RSS Feed
RSS Feed
